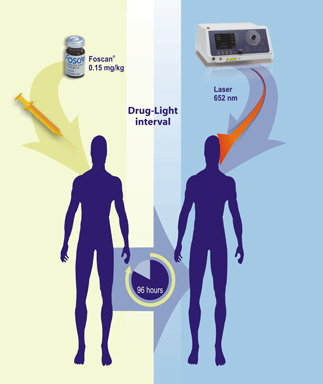
Foscan® contains temoporfin,
a photosensitizing agent, activated with non-thermal light
at a wavelength of 652 nm delivered by a laser after 4 days
of intravenous administration of the drug.
Foscan® has received Indian marketing
autorization for the palliative treatment of patients with
advanced squamous cell carcinoma of the head and neck who
have failed prior therapies and are unsuitable for radiotherapy,surgery
or systemic chemotherapy.
Foscan® is marketed in India through
Advanced Clinical Research (P) Ltd.
|